- WORKING AT A BLUE-COLLAR JOB CAN MAKE YOU A BETTER DOCTOR - 18 May 2026
- AI IN MEDICINE –IT’S IN YOUR POCKET - 30 Apr 2026
- THE PITT: 10 MEDICAL DETAILS THE SHOW GETS WRONG, AND 10 DETAILS THEY GET RIGHT - 2 Apr 2026
Artificial intelligence in medicine is coming. Will robots replace anesthesiologists?
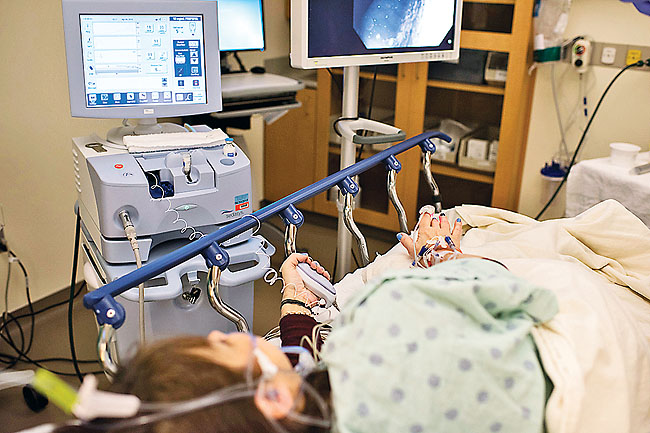
I am the Medical Director of a surgery center in California that does 5,000 gastroenterology endoscopies per year. Recently a national marketing firm contacted me to seek my opinion regarding an automated device to infuse propofol. The device was envisioned as a tool for gastroenterologist/nursing teams to use to administer propofol safely for endoscopy procedures on ASA class I – II patients.
The device was probably the SEDASYS®-Computer-Assisted Personalized Sedation System, (pictured above) developed by the Ethicon Endo-Surgery, Inc., a division of Johnson and Johnson. The SEDASYS System is a computer-assisted personalized sedation system integrating propofol delivery with patient monitoring. The system incorporates standard ASA monitors, including end-tidal CO2, into an automated propofol infusion device.
The SEDASYS system is marketed as a device to provide conscious sedation. It will not provide deep sedation or general anesthesia.
Based on pharmacokinetic algorithms, the SEDASYS infuses an initial dose of propofol (typically 30- 50 mg in young patients, or a smaller dose in older patients) over 3 minutes, and then begins a maintenance infusion of propofol at a pre-programmed rate (usually 50 mcg/kg/min). If the monitors detect signs of over- sedation, e.g. falling oxygen saturation, depressed respiratory rate, or a failure of the end-tidal CO2 curve, the propofol infusion is stopped automatically. In addition, the machine talks to the patient, and at intervals asks the patient to squeeze a hand-held gripper device. If the patient is non-responsive and does not squeeze, the propofol infusion is automatically stopped.
As of February, 2013, the SEDASYS system was not FDA approved. On May 3, 2013, Ethicon Endo-Surgery, Inc. announced that the Food and Drug Administration (FDA) granted Premarket Approval for the SEDASYS® system, a computer-assisted personalized sedation system. SEDASYS® is indicated “for the intravenous administration of 1 percent (10 milligrams/milliliters) propofol injectable emulsion for the initiation and maintenance of minimal to moderate sedation, as identified by the American Society of Anesthesiologists Continuum of Depth of Sedation, in adult patients (American Society of Anesthesiologists physical status I or II) undergoing colonoscopy and esophagoduodenoscopy procedures.” News reports indicate that SEDASYS® is expected to be introduced on a limited basis beginning in 2014.
Steve Shaffer, M.D., Ph.D., Stanford Adjunct Professor, editor-in-chief of Anesthesia & Analgesia, and Professor of Anesthesiology at Columbia University, worked with Ethicon since 2003 on the design, development and testing of the SEDASYS System both as an investigator and as chair of the company’s anesthesia advisory panel.
Dr. Shafer has been quoted as saying, “The SEDASYS provides an opportunity for anesthesiologists to set up ultra-high throughput gastrointestinal endoscopy services, improve patient safety, patient satisfaction, endoscopist satisfaction and reduce the cost per procedure.” (Gastroenterology and Endoscopy News, November 2010, 61:11)
In Ethicon’s pivotal study supporting SEDASYS, 1,000 ASA class I to III adults had routine colonoscopy or esophagogastroduodenoscopy, and were randomized to either sedation with the SEDASYS System (SED) or sedation with each site’s current standard of care (CSC) i.e. benzodiazepine/opioid combination. The reference for this study is Gastrointest Endosc. 2011 Apr;73(4):765-72. Computer-assisted personalized sedation for upper endoscopy and colonoscopy: a comparative, multicenter randomized study. Pambianco DJ, Vargo JJ, Pruitt RE, Hardi R, Martin JF.
In this study, 496 patients were randomized to SED and 504 were randomized to CSC. The area under the curve of oxygen desaturation was significantly lower for SED (23.6 s·%) than for CSC (88.0 s·%; P = .028), providing evidence that SEDASYS provided less over-sedation than current standard of care with benzodiazepine/opioid. SEDASYS patients were significantly more satisfied than CSC patients (P = .007). Clinician satisfaction was greater with SED than with CSC (P < .001). SED patients recovered faster than CSC patients (P < .001). The incidence of adverse events was 5.8% in the SED group and 8.7% in the CSC group.
Donald E. Martin, MD, associate dean for administration at Pennsylvania State Hershey College of Medicine and chair of the Section on Clinical Care at the American Society of Anesthesiologists (ASA), expressed concerns about the safety of the device. Dr. Martin (Gastroenterology and Endoscopy News, November 2010, 61:11) was quoted as saying, “SEDASYS is requested to provide minimal to moderate sedation and yet the device is designed to administer propofol in doses known to produce general anesthesia.”
Dr. Martin added that studies to date have shown that some patients who had propofol administered by SEDASYS experienced unconsciousness or respiratory depression (Digestion 2010;82:127-129, Maurer WG, Philip BK.). In the largest prospective, randomized trial evaluating the safety of the device compared with the current standard of care, five patients (1%) experienced general anesthesia with SEDASYS. The ASA also voiced concern that SEDASYS could be used in conditions that do not comply with the black box warning in the propofol label, namely that propofol “should be administered only by persons trained in the administration of general anesthesia and not involved in the conduct of the surgical/diagnostic procedure.”
Anesthetists, emergency room doctors, and trauma helicopter nurses are trained in the administration of general anesthesia. Gastroenterologists and endoscopy nurses are almost never experts in airway management. For this reason, propofol anesthetics for endoscopy are currently the domain of anesthesiologists and nurse anesthetists.
In my phone conversation regarding the automated propofol-infusion system, I told the marketing company’s representative that in my opinion a machine that infused propofol without an airway expert present could be unsafe. The marketing consultant responded that in parts of the Northeastern United States, including New York City, many GI endoscopies are done with the assistance of an anesthesia provider administering propofol. If SEDASYS were to be approved, the devices could replace anesthesiologists.
In the current fee-for-service model of anesthesia billing, anesthesiologists and CRNA’s bill insurance companies or Medicare for their professional time. If machines replace anesthesiologists and CRNA’s, the anesthesia team cannot send a fee-for-service bill for professional time. The marketing consultant foresaw that with the advent of ObamaCare and Accountable Care Organizations, if a health care organization is paid a global fee to take care of a population rather than being paid a fee-for-service sum, then perhaps the cheapest way to administer propofol sedation for GI endoscopy would be to replace anesthesia providers with SEDASYS machines.
A planned strategy is to have gastroenterologists complete an educational course that would educate them on several issues. Key elements of the course would be: 1) anesthesiologists are required if deep sedation is required, 2) SEDASYS is not appropriate if the patient is ASA 3 or 4 or has severe medical problems, 3) SEDASYS is not appropriate if the patient has risk factors such as morbid obesity, difficult airway, or sleep apnea, and 4) airway skills are to be taught in the simulation portion of the training. Specific skills are chin life, jaw thrust, oral airway use, nasal airway use, and bag-mask ventilation. Endotracheal intubation and LMA insertion are not to be part of the class. If the endoscopist cannot complete the procedure with moderate sedation, the procedure is to be cancelled and rescheduled with an anesthesia provider giving deep IV sedation.
Some anesthesiologists are concerned about being pushed out of their jobs by nurse anesthetists. It may be that some anesthesiologists will be pushed out of their jobs by machines.
I’ve been told that the marketing plan for SEDASYS is for the manufacturer to give the machine to a busy medical facility, and to only charge for the disposable items needed for each case. The disposable items would cost $50 per case. In our surgery center, where we do 5,000 cases per year, this would be an added cost of $25,000 per year. There would be no significant savings, because we do no use anesthesiologists for most gastroenterology sedation.
There have been other forays into robotic anesthesia, including:
1) The Kepler Intubation System (KIS) intubating robot, designed to utilized video laryngoscopy and a robotic arm to place an endotracheal tube (Curr Opin Anaesthesiol. 2012 Oct 25. Robotic anesthesia: not the realm of science fiction any more. Hemmerling TM, Terrasini N. Departments of Anesthesia, McGill University),
2) The McSleepy intravenous sedation machine, designed to administer propofol, narcotic, and muscle relaxant to patients to control hypnosis, analgesia, and muscle relaxation. (Curr Opin Anaesthesiol. 2012 Dec;25(6):736-42. Robotic anesthesia: not the realm of science fiction any more. Hemmerling TM, Terrasini N.)
3) The use of the DaVinci surgical robot to perform regional anesthetic blockade. (Anesth Analg. 2010 Sep;111(3):813-6. Epub 2010 Jun 25. Technical communication: robot-assisted regional anesthesia: a simulated demonstration. Tighe PJ, Badiyan SJ, Luria I, Boezaart AP, Parekattil S.).
4) The use of the Magellan robot to place peripheral nerve blocks (Anesthesiology News, 2012, 38:8)
Each of these applications may someday lead to the performance of anesthesia by an anesthesiologist at geographical distance from the patient. In an era where 17% of the Gross National Product of the United States is already being spent on health care, one can question the logic of building expensive technology to perform routine tasks like I.V. sedation, endotracheal intubation, or regional block placement. The new inventions are futuristic and interesting, but a DaVinci surgical robot costs $1.8 million, and who knows what any of these anesthesia robots would sell for? The devices seem more inflationary than helpful at this point.
Will robots replace anesthesiologists? Inventors are edging in that direction. I would watch the peer-reviewed anesthesia journals for data that validates the utility and safety of any of these futuristic advances.
It will be a long time before anyone invents a machine or a robot that can perform mask ventilation. SEDASYS is designed for conscious sedation, not deep sedation or general anesthesia. Anyone or anything that administers general anesthesia without expertise in mask ventilation and all facets of airway management is courting disaster.
THE iCONTROL-RP ANESTHESIA ROBOT
On May 15, 2015, the Washington Post published a story titled, “We Are Convinced the Machine Can Do Better Than Human Anesthesiologists.”
Is this true? Are anesthesiologists on the verge of being replaced by a new robot?
In a word, “No.”
The new device being discussed is the iControl-RP anesthesia robot.
In recent years there have been significant advances in the automated delivery of the intravenous anesthetic drugs propofol and remifentanil. (Orliaguet GA, Feasibility of closed-loop titration of propofol and remifentanil guided by the bispectral monitor in pediatric and adolescent patients: a prospective randomized study, 2015 Apr;122(4):759-67). Propofol is an ultra-short-acting hypnotic drug that causes sleep. Remifentanil is an ultra-short-acting narcotic that relieves pain. Administered together, these drugs induce what is referred to as Total Intravenous Anesthesia, or TIVA. Total Intravenous Anesthesia is a technique anesthesiologists use when they choose to avoid using inhaled gases such as sevoflurane and nitrous oxide. Anesthesiologists administer TIVA by adjusting the flow rates on two separate infusion pumps, one infusion pump containing each drug.
A closed-loop system is a machine that infuses these drugs automatically. These systems include several essential items: The first is a processed electroencephalogram (EEG) such as a bi-spectral monitor (BIS monitor) attached to the patient’s forehead which records a neurologic measure of how asleep the patient is. The BIS monitor calculates a score between 0 and 100 for the patient’s level of unconsciousness, with a score of 100 corresponding to wide awake and 0 corresponding to a flat EEG. A score of 40 – 60 is considered an optimal amount of anesthesia depth. The second and third essential items of a closed-loop automated system are two automated infusion pumps containing propofol and remifentanil. A computer controls the infusion rate of a higher or lower amount of these drugs, depending on whether the measured BIS score is higher or lower than the 40- 60 range.
Researchers in Canada have expanded this technology into a device they call the iControl-RP, which is in clinical trials at the University of British Columbia. The iControl-RP is a closed-loop system which makes its own decisions. The initials RP stand for the two drugs being titrated: remifentanil and propofol. In addition to monitoring the patient’s EEG level of consciousness (via a BIS monitor device called NeuroSENSE), this new device monitors traditional vital signs such as blood oxygen levels, heart rate, respiratory rate, and blood pressure, to determine how much anesthesia to deliver.
Per published information on their research protocol, the iControl-RP allows either remifentanil or propofol to be operated in any of three modes: (1) closed-loop control based on feedback from the EEG as measured by the NeuroSENSE; (2) target-controlled infusion (TCI), based on previously-described pharmacokinetic and pharmacodynamic models; and (3) conventional manual infusion, which requires a weight-based dose setting. (Reference: Closed-loop Control of Anesthesia: Controlled Delivery of Remifentanil and Propofol Dates, Status, Enrollment Verified by: Fraser Health, August 2014, First Received: January 15, 2013, Last Updated: March 5, 2015, Phase: N/A, Start Date: February 2013, Overall Status: Recruiting, Estimated Enrollment: 150).
In Phase 1 of the iControl-RP testing involving 50 study subjects, propofol will be administered in closed-loop mode and a remifentanil infusion will be administered based on a target-controlled infusion. In phase 2 involving 100 study subjects, both propofol and remifentanil will be administered in closed-loop mode. The investigators aim to demonstrate that closed-loop control of anesthesia and analgesia based on EEG feedback is clinically feasible.
In both phases, an anesthesiologist will monitor the patient as per routine practice and have the ability to modify the anesthetic or analgesic drugs being administered. That is, he or she will be able to adjust the target depth of hypnosis, adjust the target effect site concentration for remifentanil, immediately switch to manual control of either infusion, administer a bolus dose, or immediately stop the infusion of either drug. iControl-RP is connected to the NeuroSENSE EEG monitor, the two infusion pumps for separately controlled propofol and remifentanil administration, and the operating room patient vital signs monitor. A user interface allows the anesthesiologist to set the target EEG depth level, switch between modes of operation (manual, target-controlled infusion, or closed-loop), and set manual infusion rates or target effect-site concentrations for either drug as required.
Per the article in the Washington Post. (Todd C. Frankel, Washington Post, May 15, 2015), one of the machine’s co-developers Mark Ansermino, MD said, “We are convinced the machine can do better than human anesthesiologists.” The iControl-RP has been used to induce deep sedation in adults and children undergoing general surgery. The device had been used on 250 patients so far.
Why is this robotic device only a small step toward replacing anesthesiologists?
A critical realization is that anesthetizing patients requires far more skill than merely titrating two drug levels. Every patient requires (1) preoperative assessment of all medical problems from the history, physical exam, and laboratory evaluation of each individual patient, so that the anesthesiologist can plan and prescribe the appropriate anesthesia type; (2) placement of an intravenous line through which the TIVA drugs may be administered; (3) mask ventilation of an unconscious patient (in most cases), followed by placement of an airway tube to control the delivery of oxygen and ventilation in and out of the patient’s lungs; (4) observation of all vital monitors during surgery, with the aim of directing the diagnosis and treatment of any complication that occurs as a result of anesthesia or the surgical procedure; (5) removal of the airway tube at the conclusion of most surgeries, and (6) the diagnosis and treatment of any complication in the newly awake patient following the anesthetic.
In the future, closed-loop titration of drugs may lessen an anesthesiologist’s workload and free him or her for other activities. In the distant future, closed-loop titration of drugs may free a solitary anesthesiologist to initiate and monitor multiple anesthetics simultaneously from a control booth via multiple video screens and interface displays. But the handling of all tasks (1) – (6) by an automated robotic device is still the stuff of science fiction. The Washington Post article said an early role for the machine could be in war zones or remote areas where an anesthesiologist is unavailable. One could conjecture that a closed-loop anesthesia system may be used to facilitate surgery in outer space some day as well.
In either case, an anesthesiologist or some other highly-trained medical professional will still be required on site to achieve tasks (1) – (6).
The iControl-RP has not been approved by the U.S. Food and Drug Administration.
The iControl-RP team has struggled to find a corporate backer for its project. Dr. Ansermino, the anesthesiologist inventor in Vancouver, told the Washington Post, “Most big companies view this as too risky,” but he believed a device like this was inevitable. “I think eventually this will happen,” Ansermino told the Washington Post, “whether we like it or not.”
That may be, but I suspect companies are risk averse regarding the iControl-RP because investment is guided by analysts and physicians who must consider the practical applications and risks of any new medical device. The issues of leaving (1) – (6) up to a robotic device are impractical at best, and dangerous to the patient at worse.

I’m fascinated by the topic of artificial intelligence (AI). This is the third column in a series regarding robots in medicine. (See Robot Anesthesia and Robot Anesthesia II)
AI already influences our daily life. Smartphones verbally direct us to our destination through mazes of highways and traffic. Computers analyze our shopping habits and populate our Internet screens with advertisements for products we’ve ogled in the past. Smartphones perform voice-to-text conversions by pattern recognition of human vocal sounds. Fingerprint scanners learn and then recognize the image of our thumbprints with exacting accuracy. Amazon’s Alexa is an AI-powered personal assistant that accepts verbal commands in our homes.
What about artificial intelligence in medicine (AIM)? AIM is a bold enterprise on the horizon in clinical medicine. Hundreds of AIM scientific publications appear in medical journals each year. I’m not an AIM researcher, but I’m an expert clinician and I love to read. I’ve worked in almost every scenario of medical practice, and because my base is at Stanford University Medical Center in Silicon Valley, many of the advances of the high-tech industry are right here in my backyard. My medical board certifications are in internal medicine and anesthesiology—two fields which have significant overlap in their knowledge base but radically different practice settings. Internal medicine doctors work in clinics, where most diseases are chronic and the most valuable tools for doctors are excellent listening and diagnostic skills. Anesthesiologists work in operating rooms and intensive care units—acute care settings which demand vigilance, steady hands, and quick thinking.
Based on my experience and my reading, I foresee AIM/robots populating three clinical arenas in radically different roles. These arenas will be: 1) diagnosis of images, 2) clinics, and 3) operating rooms/intensive care units. Let’s look at each of these in turn.
- Diagnosis of images This will be the first major application of AIM. We already have electrocardiogram (ECG) machines which interpret a patient’s ECG tracing with high accuracy, and print out the diagnosis for the physician to read. This application debuted in the 1980s and is now the industry standard, although confirmation of diagnosis by a physician is important for some diagnoses such as ST-elevation myocardial infarction (STEMI). More than a few physicians have already lost the skill of reading an ECG themselves because of this device. Future applications of image analysis in medicine will be machine learning for diagnosis in radiology, pathology, and dermatology. The evaluation of digital X-rays, MRIs, or CT scans is the assessment of arrays of pixels. Expect that future computer programs will be as accurate or more accurate than human radiologists. The model for machine learning is similar to the fashion in which a human child learns. A child is not given a list of criteria which define what a dog looks like. Instead, the child sees an animal and his parents tell him that animal is a dog. After repeated exposures, the child learns what a dog looks like. Early on the child may be fooled into thinking that a wolf is a dog, but with increasing experience the child can discern with almost perfect accuracy what is or is not a dog. Machine learning is a subset of deep learning, a concept that makes automated decision-making possible. Deep learning is a radically different method of programming computers. It requires massive database entry, much like the array of dogs that a child sees in the example above, so that the computer can learn the skill of pattern matching. The program repetitively teaches a machine the identity of certain images, and the system hones this algorithm and becomes faster and more accurate in recognizing similar images. An AI computer which masters machine learning and deep learning will probably not give yes or no answers, but rather a percentage likelihood of a diagnosis, i.e. a radiologic image has greater than a 99% chance of being normal, or a skin lesion has greater than a 99% chance of being a malignant melanoma. At the present time the Food and Drug Administration (FDA) does not allow machines to make formal diagnoses, and such AI computer applications are only prototypes. But if you’re a physician who makes his or her living by interpreting digital images, there’s real concern about AI taking your job in the future. Some experts believe AIM devices will not replace radiologists, but rather will make their work more efficient and accurate. For example, AI computers can identify MRI or CT scans which are normal, freeing human radiologists to concentrate on scans where an abnormality exists. In this scenario, radiologists would not lose their jobs to AIM computers, instead radiologists who don’t use AIM machines may lose their jobs to radiologists who do use the AIM technology. In pathology, computerized digital diagnostic skills will be applied to microscopic diagnosis. In dermatology, machine learning will be used to diagnosis skin cancers, based on large learned databases of digital photographs. Dermatologists must rely on years of experience to learn to discern various skin lesions, but an AI computer can ingest hundreds of thousands of images in a period of months.
- Clinics In the clinic setting, the desired AI application would be a computer that could input information on a patient’s history, physical examination, and laboratory studies, and via machine learning and deep learning, establish the patient’s diagnoses with a high percentage of success. AI computers will be stocked with information from multiple sources, including all known medical knowledge published in textbooks and journals, as well as the electronic health records (EHR)/ clinical data from thousands of previous hospital and clinic patients. AI machines can remember this vast array of information better than any human physician. AI machines will organize the input of new patient information into a flowchart, also known as a branching tree. A flowchart will mimic the process a physician carries out when asking a patient a series of questions. The flowchart program contains a series of “if . . . then . . .” branches that depend on the patient’s answers. AI will input the information sources from each new patient, and arrive at diagnoses. Once each diagnosis is established with a reasonable degree of medical certainty, an already-established algorithm for treatment of that diagnosis can be applied. For example, if the computer makes a diagnosis of asthma, then an established textbook treatment regimen of bronchodilators will be activated. It’s projected that AIM applications in clinic settings will decrease unnecessary diagnostic tests, lower therapeutic costs, and reduce the manpower needed for outpatient medicine.
- Operating rooms The best current example of robot technology in the operating room is the da Vinci operating robot, used primarily in urology and gynecologic surgery. This robot is not intended to have an independent existence, but rather enables the surgeon to see inside the body in three dimensions and to perform fine motor procedures at a higher level. In my previous essays Robot Anesthesia and Robot Anesthesia II, I described models of robots designed to perform intravenous sedation or intubation of the trachea, products which are futuristic but currently have no market share. The good news for procedural physicians such as anesthesiologists or surgeons is this: it’s unlikely any AI computer or robot will be able to independently replace the manual skills such as airway management, endotracheal intubation, or surgical excision. Regarding anesthesiology, I expect future AIM robots will be hyperattentive monitoring devices which follow the vital signs of anesthetized patients, and then utilize feedback loops to titrate or adjust the depth of anesthetic drugs as indicated by these vital signs. Such a robot would not replace a human anesthesiologist, but could serve as an autopilot analogue during the maintenance or middle phase of long anesthetics, freeing up the anesthesia professional so that he or she need not be physically present. This parallels the original genesis of the role of a nurse anesthetist—to be present during stable phases of anesthetic management—so that the physician anesthesiologist could roam to other operating rooms as needed.
What will an AIM robot doctor look like? It’s unlikely it will look like a human. Most sources project it will look like a smartphone. I’d expect the screen to be bigger than a smartphone screen, so an AIM robot doctor will likely look like a tablet computer. For certain applications such as clinic diagnosis or new image retrieval, the AIM robot will have a camera, perhaps on a retractable arm so that the camera can approach various aspects of a patient’s anatomy as indicated. Individual patients will need to sign in to the computer software system—this will be done via tools such as retinal scanners, fingerprint scanners, or face recognition programs—so that the computer can retrieve that individual patient’s EHR data from an Internet cloud. It’s possible individual patients will be issued a card, not unlike a debit or credit card, which includes a chip linking them to their EHR data.
How will we define if these medical computers are truly intelligent? The accepted test for machine intelligence is the Turing test, as described by computer scientist Alan Turing in 1950. In the Turing test, a human evaluator interacts with two players via a computer keyboard. One of the players is a human and the other a machine. If the evaluator cannot reliably tell the machine from the human, the machine is said to have passed the test, and is deemed intelligent.
What will be the economics of AIM? Who will pay for it? Currently America spends 17.6% of its Gross National Product on healthcare, and this number is projected to reach 20% by 2025. Entrepreneurs realize that healthcare is a multi-billion dollar industry, and the opportunity to earn those healthcare dollars is a seductive lure. Companies are looking to merge increasing computing power available at steadily decreasing costs, big data from large EHR patient populations, and artificial intelligence with an aim to drive down the costs of health care while increasing effectiveness. Expect to see the development of increasingly cheaper AIM devices to augment the skills of human physicians, or maybe replace them in some job descriptions. The government’s medical costs may decrease if work currently done by expensive-to-train physicians is instead performed by nurse practitioners or nurses aided by artificial intelligence machines, supervised by relatively few human physicians. Google is working on an AIM project in the United Kingdom entitled DeepMind. DeepMind is using machine learning to analyze eye scans from more than a million patients, with the aim to create algorithms which can detect early warning signs of eye diseases that human physicians might miss. Google researchers have also developed an AIM computer to screen for and analyze the spread of breast cancer cells in lymph node tissue on pathology slide images. Scientists at the Memorial Sloan-Kettering Cancer Center in New York have programmed over 600,000 medical evidence reports, 1.5 million patient medical records, and two millions of pages of text from medical journals into IBM’s Watson computer. Equipped with more information than any human physician could ever remember, Watson is projected to become a diagnostic machine superior to any doctor.
There’s a worldwide shortage of physicians. The earliest a human physician can enter the workforce is age 29, after completing 4 years of college, 4 years of medical school, and 3 years of the shortest residency (e.g. internal medicine, pediatrics, or family practice residency). A major advantage of AIM is that the machines won’t require 24 years of education. Can America afford to train people for almost three decades to then sit in a clinic and perform histories and physicals on patients who have chronic illnesses such as hypertension, hyperlipidemia, and obesity? Shifting these jobs to allied healthcare providers such as physician assistants or nurse practitioners is a cheaper alternative, but what could be cheaper than an AIM machine module which either assists one physician to evaluate a vast number of patients, or an AIM module of the future which replaces the physician entirely?
When can we expect to see new AIM tools adopted in clinical practice? Web-based smartphone apps such as Your.MD and Babylon already exist to assist physicians in diagnosis. You can anticipate the application of machine learning in the diagnosis of digital images soon. The DeepMind and Watson computers are blazing a trail toward machine learning in clinical medicine. Expect the FDA to assess the new technologies, and when it is safe and appropriate, to approve machine diagnosis as part of the practice of medicine. Remember how fast we advanced from a cell phone the size of a breadbox to the powerful smartphone that fits in the palm of your hand today. In the ten years since the introduction of the iPhone in 2007, who could have imagined the vast array of applications we carry in our pocket or purse in 2017?
AIM is coming. It will arrive be sooner than we think, and in all likelihood it will be more powerful and more wonderful than we could imagine. A brave prediction: AIM will change medicine more than any development since the invention of anesthesia in 1849.
I can’t wait to see it.
Recommended reading:
Hsieh, Paul. AI in Medicine: Rise of the Machines, Forbes, April 30, 2017.
Omni staff. Artificial Intelligence in Medicine. Omni, 2016.
Dickson B. How Artificial Intelligence is Revolutionizing Healthcare, TheNextWeb.com, May 2017.
Russell S, Norvig P. Arificial Intelligence, A Modern Approach, 3rd Edition, 2010, Prentice Hall.
*
*
*
*
Published in September 2017: The second edition of THE DOCTOR AND MR. DYLAN, Dr. Novak’s debut novel, a medical-legal mystery which blends the science and practice of anesthesiology with unforgettable characters, a page-turning plot, and the legacy of Nobel Prize winner Bob Dylan.
KIRKUS REVIEW
In this debut thriller, tragedies strike an anesthesiologist as he tries to start a new life with his son.
Dr. Nico Antone, an anesthesiologist at Stanford University, is married to Alexandra, a high-powered real estate agent obsessed with money. Their son, Johnny, an 11th-grader with immense potential, struggles to get the grades he’ll need to attend an Ivy League college. After a screaming match with Alexandra, Nico moves himself and Johnny from Palo Alto, California, to his frozen childhood home of Hibbing, Minnesota. The move should help Johnny improve his grades and thus seem more attractive to universities, but Nico loves the freedom from his wife, too. Hibbing also happens to be the hometown of music icon Bob Dylan. Joining the hospital staff, Nico runs afoul of a grouchy nurse anesthetist calling himself Bobby Dylan, who plays Dylan songs twice a week in a bar called Heaven’s Door. As Nico and Johnny settle in, their lives turn around; they even start dating the gorgeous mother/daughter pair of Lena and Echo Johnson. However, when Johnny accidentally impregnates Echo, the lives of the Hibbing transplants start to implode. In true page-turner fashion, first-time novelist Novak gets started by killing soulless Alexandra, which accelerates the downfall of his underdog protagonist now accused of murder. Dialogue is pitch-perfect, and the insults hurled between Nico and his wife are as hilarious as they are hurtful: “Are you my husband, Nico? Or my dependent?” The author’s medical expertise proves central to the plot, and there are a few grisly moments, as when “dark blood percolated” from a patient’s nostrils “like coffee grounds.” Bob Dylan details add quirkiness to what might otherwise be a chilly revenge tale; we’re told, for instance, that Dylan taught “every singer with a less-than-perfect voice…how to sneer and twist off syllables.” Courtroom scenes toward the end crackle with energy, though one scene involving a snowmobile ties up a certain plot thread too neatly. By the end, Nico has rolled with a great many punches.
Nuanced characterization and crafty details help this debut soar.
Click on the image below to reach the Amazon link to The Doctor and Mr. Dylan:
LEARN MORE ABOUT RICK NOVAK’S FICTION WRITING AT RICK NOVAK.COM BY CLICKING ON THE PICTURE BELOW:




One thought on “ROBOT ANESTHESIA”